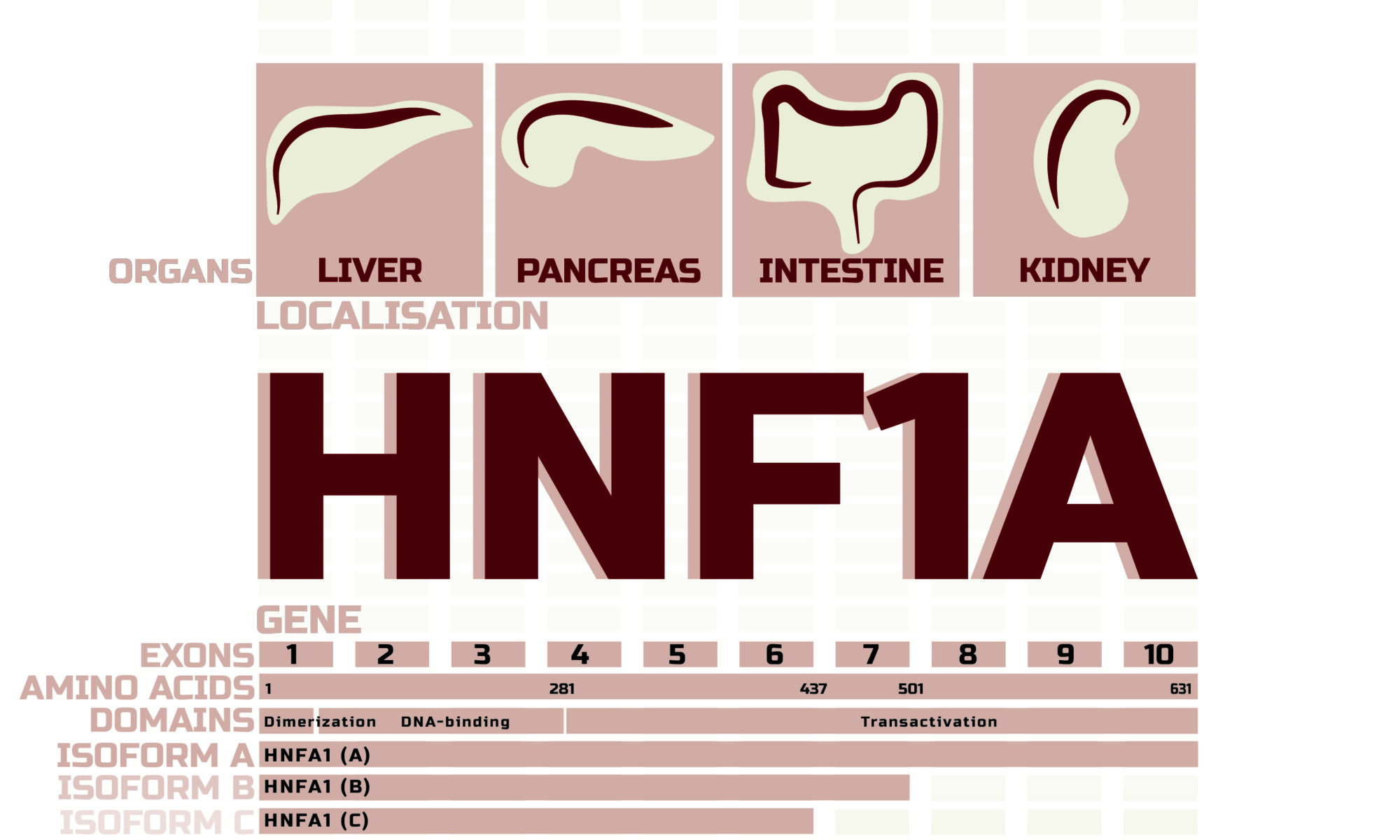
Maturity-onset diabetes of the young (MODY) represents an autosomal dominant genetic form of diabetes, coined by Tattersall and Fajans in 1974 as having one or more variations in a single gene . MODY is initially characterised by fasting hyperglycaemia, often diagnosed in children or young adults. Currently, MODY accounts for 1–5% of all individuals with diabetes, and at least 14 distinct subtypes have been reported to date. Heterozygous mutations in the gene encoding hepatocyte nuclear factor 1-alpha (HNF1A) cause the most common form of monogenic diabetes, known as HNF1A-MODY. This form of MODY is highly prevalent in populations with European ancestry (>70%) and is more frequent than previously thought. Approximately 95% of individuals with HNF1A-MODY are misdiagnosed as either type 1 diabetes or type 2 diabetes, thus revealing a lack of physician awareness and/or access to genetic testing . Early diagnosis is critical since HNF1A-MODY affects 50% of children born to a mutation carrier, and penetrance is often above 90%, such that three or more generations are usually affected.
Institut Pasteur de Lille welcomes students
The Institut Pasteur de Lille has an international medical research center on its campus divided into eight units, with multidisciplinary and transversal skills.
DiabInnov
DiabInnov is a research platform focused on innovative strategies for the treatment of metabolic disorders and their comorbidities to provide significant advance in patient care.
How to be a good science communicator | Nature Medicine
During the COVID-19 pandemic, the public have turned to scientists and doctors for vital information. But science communication is a skill in itself, so we asked the experts to share their tips. Source : How to be a good science communicator | Nature Medicine
Source : How to be a good science communicator | Nature Medicine
Probing the interactions between air bubbles and (bio)interfaces at the nanoscale using FluidFM technology


Understanding the molecular mechanisms underlying bubble-(bio)surfaces interactions is currently a
challenge that if overcame, would allow to understand and control the various processes in which they
are involved. Atomic force microscopy is a useful technique to measure such interactions, but it is limited
by the large size and instability of the bubbles that it can use, attached either on cantilevers or on surfaces.
We here present new developments where microsized and stable bubbles are produced using
FluidFM technology, which combines AFM and microfluidics. The air bubbles produced were used to
probe the interactions with hydrophobic samples, showing that bubbles in water behave like hydrophobic
surfaces. They thus could be used to measure the hydrophobic properties of microorganisms’ surfaces,
but in this case the interactions are also influenced by electrostatic forces. Finally a strategy was developed
to functionalize their surface, thereby modulating their interactions with microorganism interfaces.
This new method provides a valuable tool to understand bubble-(bio)surfaces interactions but also to
engineer them.
Irem Demir a,b, Ines Lüchtefeld c, Claude Lemen a, Etienne Dague b,d, Pascal Guiraud a,d, Tomaso Zambelli c, Cécile Formosa-Dague
https://www.sciencedirect.com/science/article/abs/pii/S0021979721010961?via%3Dihub
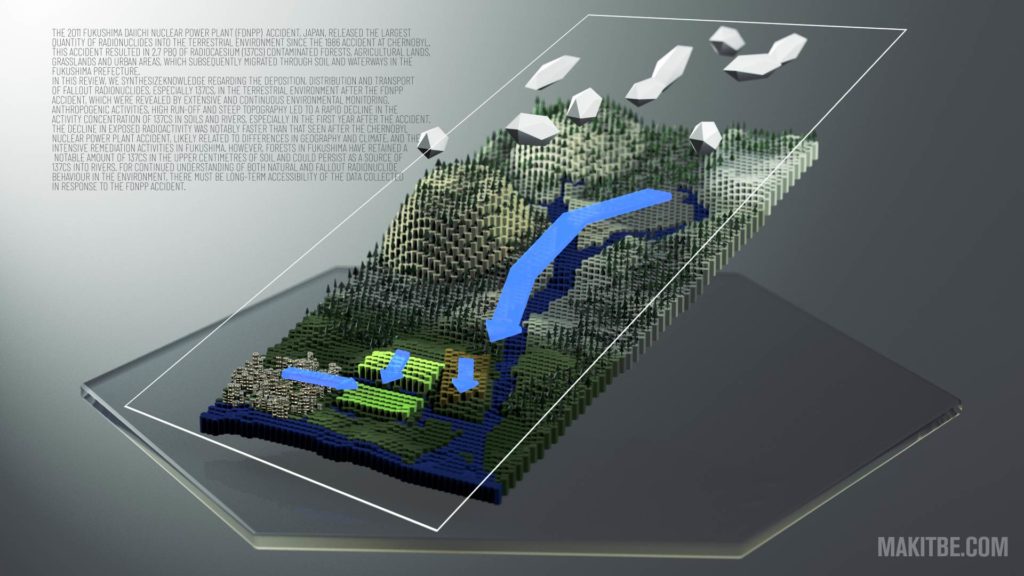
Radionuclides from the Fukushima Daiichi Nuclear Power Plant in terrestrial systems
Abstract
The 2011 Fukushima Daiichi Nuclear Power Plant (FDNPP) accident, Japan, released the largest quantity of radionuclides into the terrestrial environment since the 1986 accident at Chernobyl. This accident resulted in 2.7 PBq of radiocaesium (137Cs) contaminated forests, agricultural lands, grasslands and urban areas, which subsequently migrated through soil and waterways in the Fukushima Prefecture. In this Review, we synthesize knowledge regarding the deposition, distribution and transport of fallout radionuclides, especially 137Cs, in the terrestrial environment after the FDNPP accident, which were revealed by extensive and continuous environmental monitoring. Anthropogenic activities, high run-off and steep topography led to a rapid decline in the activity concentration of 137Cs in soils and rivers, especially in the first year after the accident. The decline in exposed radioactivity was notably faster than that seen after the Chernobyl Nuclear Power Plant accident, likely related to differences in geography and climate, and the intensive remediation activities in Fukushima. However, forests in Fukushima have retained a notable amount of 137Cs in the upper centimetres of soil and could persist as a source of 137Cs into rivers. For continued understanding of both natural and fallout radionuclide behaviour in the environment, there must be long-term accessibility of the data collected in response to the FDNPP accident.
https://www.nature.com/articles/s43017-020-0099-x?fbclid=IwAR0_wypUbVbexzpVTYO4GdI9jqJTmSFlt04gspMMSdSrrJ3wYRsuKI-UxFk#auth-Yuichi-Onda





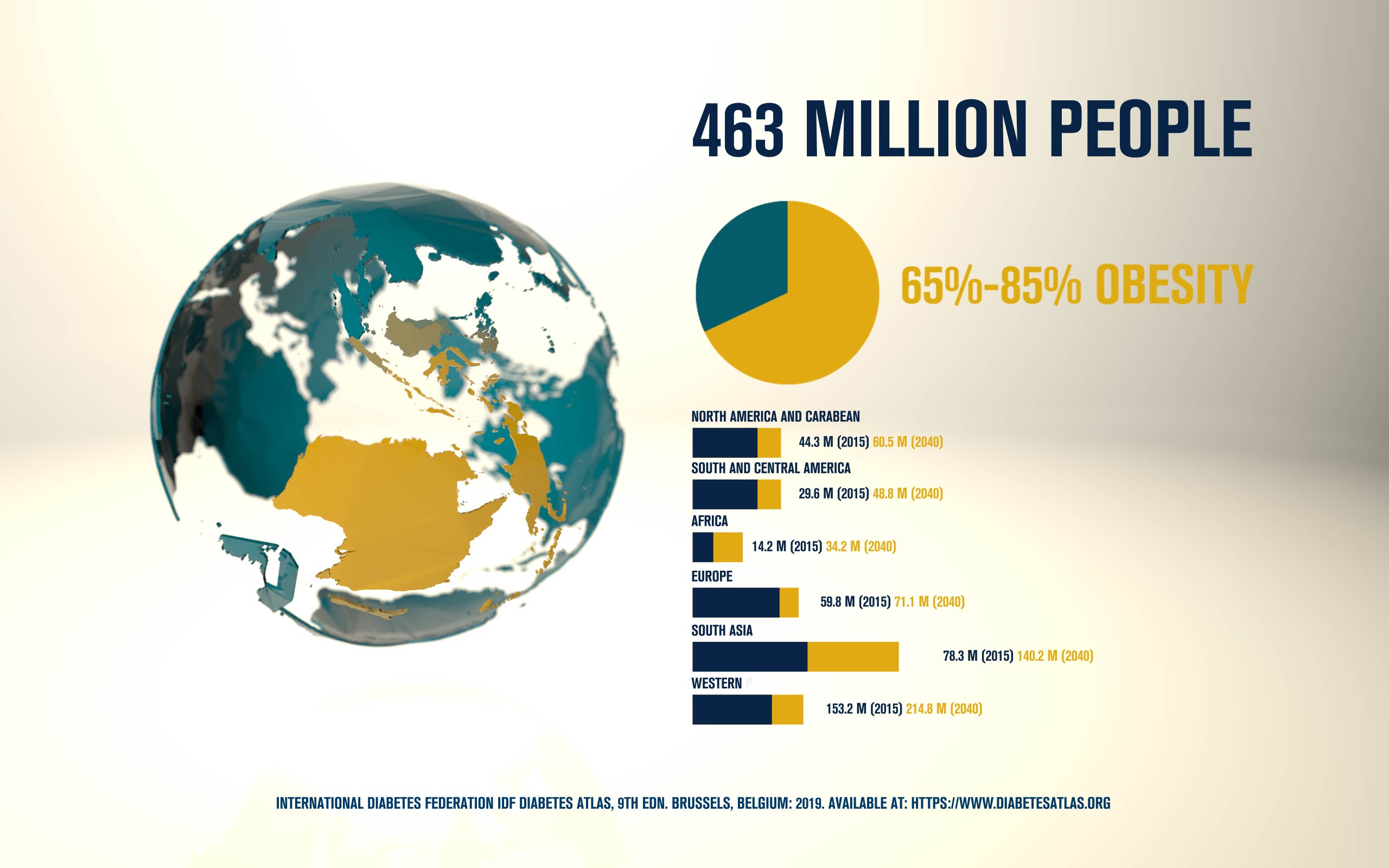
Australasian Diabetes Congress 2020 – SGLT2 and Glucagon secretion by Caroline Bonner (Pasteur Lille)
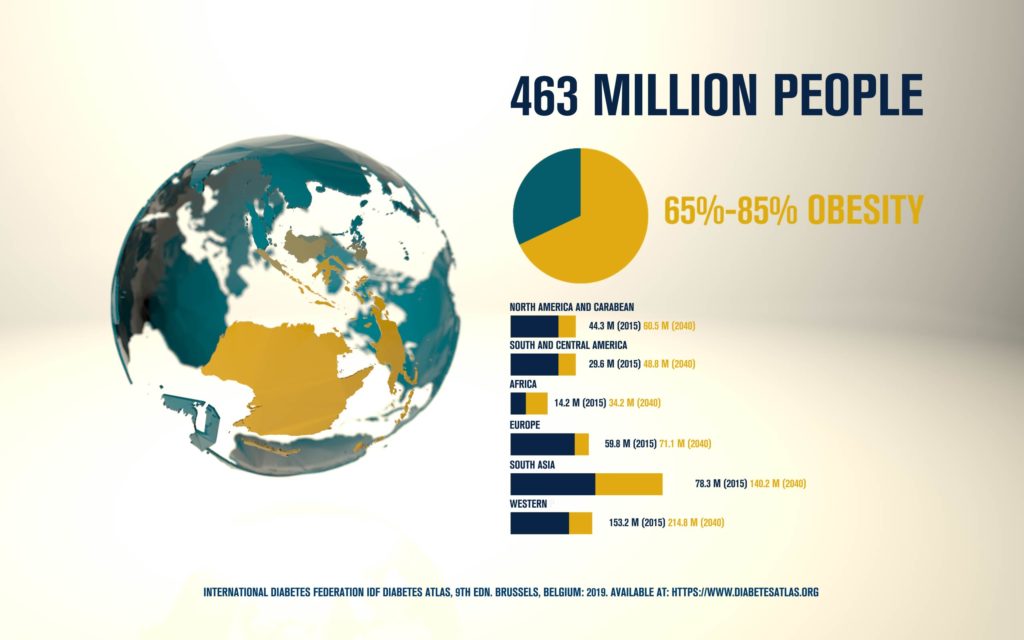
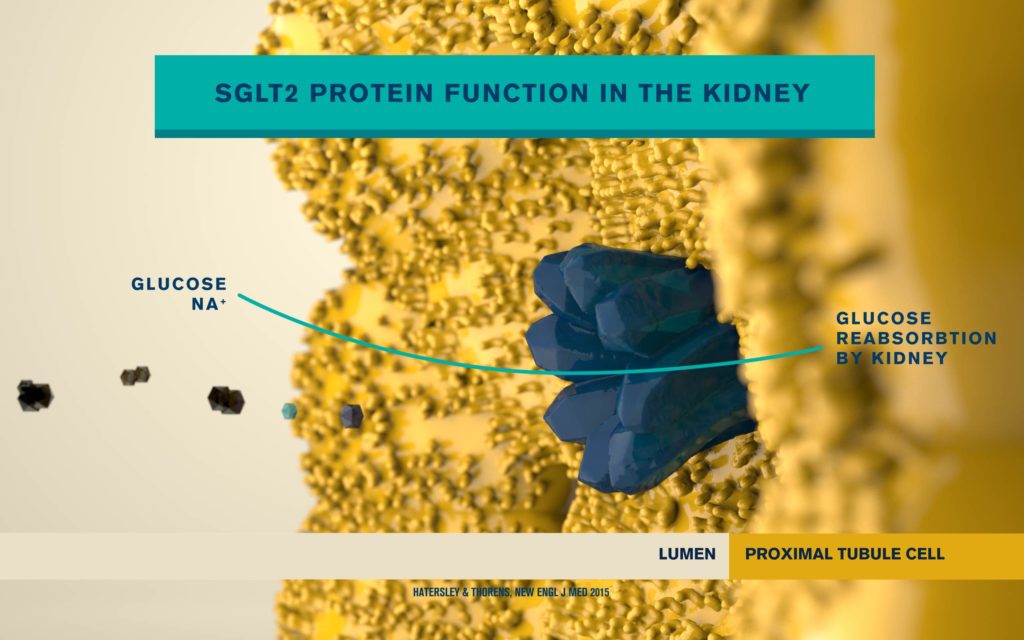
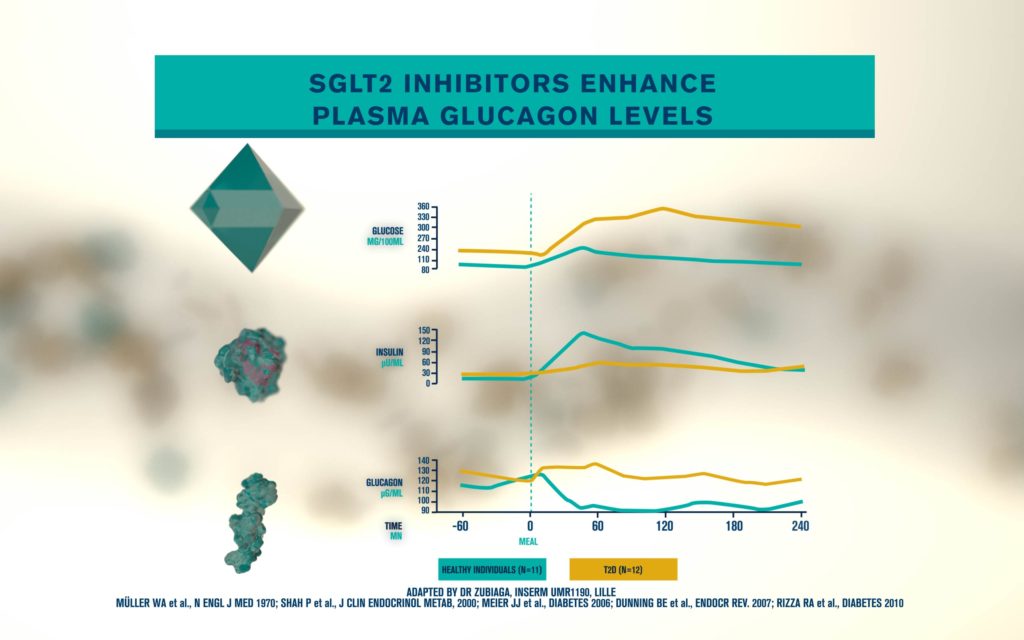
According to the International Diabetes Federation, 463 million people are living with diabetes. Obesitycontributes to 65-80% of new cases of type 2 diabetes (T2D). T2D is a complex metabolic disorder, the pathogenesis of which is not understood. Impaired insulin sensitivity, hyperglucagonemia, endogenous glucose production (EGP), and islet cell dysfunction are major traits of the disease, but the sequence of events leading to diabetes is much more complex than we expected. For decades, the mechanisms by which nutrients stimulate insulin secretion have been studied extensively, whilst the neighboring alpha cells (which secrete glucagon) have been somewhat ignored. A heightened interest in the regulation of glucagon secretion became apparent when T2D patients treated with Sodium-Glucose Co-Transporter-2 (SGLT2) inhibitors presented with increased plasma glucagon levels and EGP, a phenomenon that later attracted attention to the heterogeneous expression and function of SGLT2 in human alpha cells (Bonner et al., Nature Medicine 2015; Saponaro et al., Diabetes 2020). This concerning side effect, in a patient population already affected by hyperglucagonemia, prompted us to investigate whether the glucagon-lowering actions of the glucagon-like peptide 1 receptor agonist liraglutide could reduce the glucagon over-secretion induced by the SGLT2 inhibitor dapagliflozin. Indeed, we found that dual treatment with dapagliflozin and liraglutide
produced a sustainable reduction of glycemia compared with each drug alone. Moreover, liraglutide reduced dapagliflozin-induced glucagon secretion via somatostatin release, without any significant change in insulin secretion from human beta cells in culture (Saponaro et al., Cell Reports, 2019). By contrast, native GLP1 induced insulin secretion at low picomolar concentrations under the same experimental conditions.
Unexpectedly, we found in vivo in mice that the effects of liraglutide on insulin secretion were regulated by the central nervous system (unpublished data). Together these findings revealed that human islet cultures represent a valuable commodity to complement studies in vivo to obtain an overall view of the several potential mechanisms of these drugs.
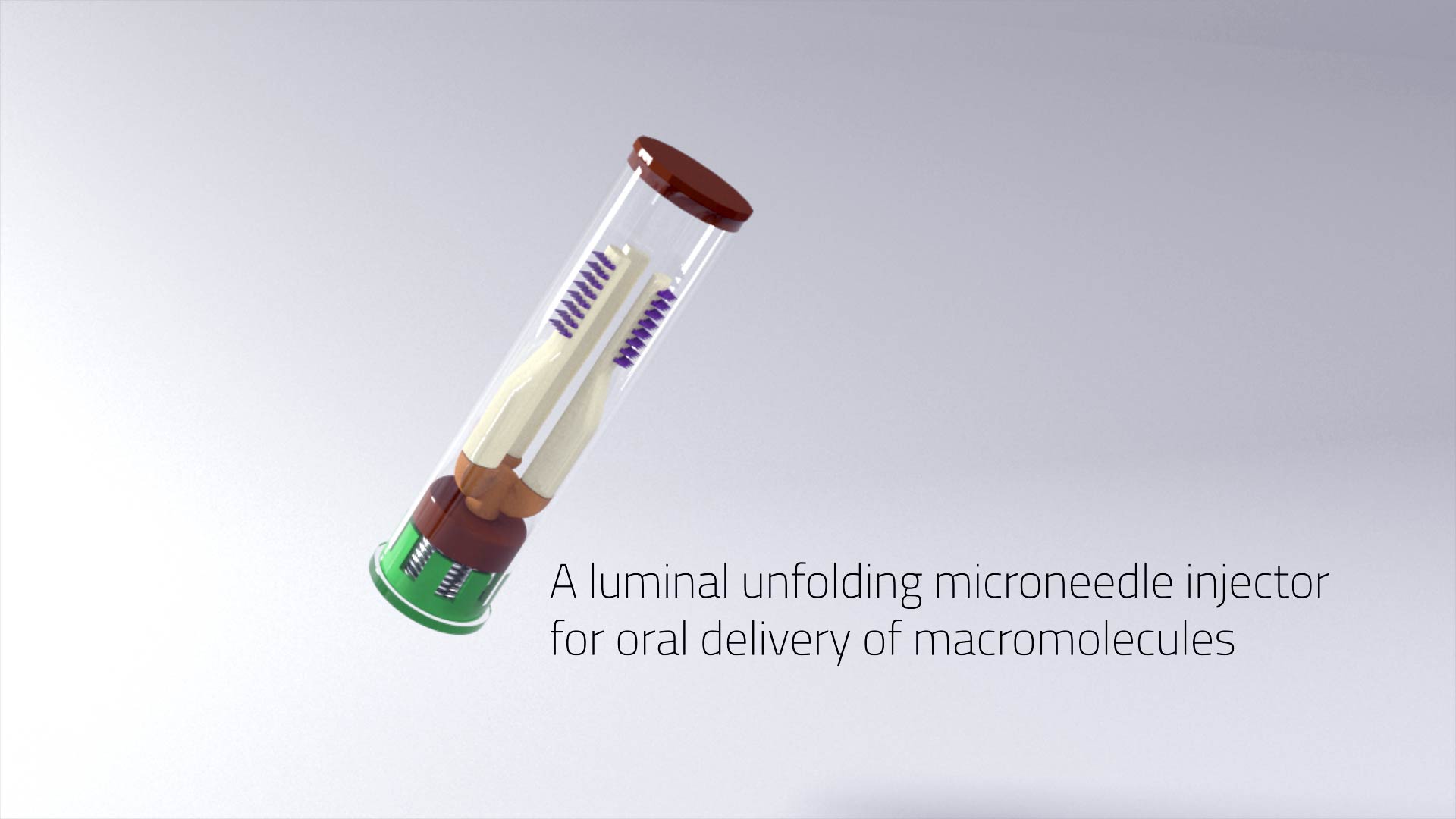
A luminal unfolding microneedle injector for oral delivery of macromolecules – Nature Medicine
“A luminal unfolding microneedle injector for oral delivery of macromolecules”
Abstract
Insulin and other injectable biologic drugs have transformed the treatment of patients suffering from diabetes1,2, yet patients and healthcare providers often prefer to use and prescribe less effective orally dosed medications3,4,5. Compared with subcutaneously administered drugs, oral formulations create less patient discomfort4, show greater chemical stability at high temperatures6, and do not generate biohazardous needle waste7. An oral dosage form for biologic medications is ideal; however, macromolecule drugs are not readily absorbed into the bloodstream through the gastrointestinal tract8. We developed an ingestible capsule, termed the luminal unfolding microneedle injector, which allows for the oral delivery of biologic drugs by rapidly propelling dissolvable drug-loaded microneedles into intestinal tissue using a set of unfolding arms. During ex vivo human and in vivo swine studies, the device consistently delivered the microneedles to the tissue without causing complete thickness perforations. Using insulin as a model drug, we showed that, when actuated, the luminal unfolding microneedle injector provided a faster pharmacokinetic uptake profile and a systemic uptake >10% of that of a subcutaneous injection over a 4-h sampling period. With the ability to load a multitude of microneedle formulations, the device can serve as a platform to orally deliver therapeutic doses of macromolecule drugs.
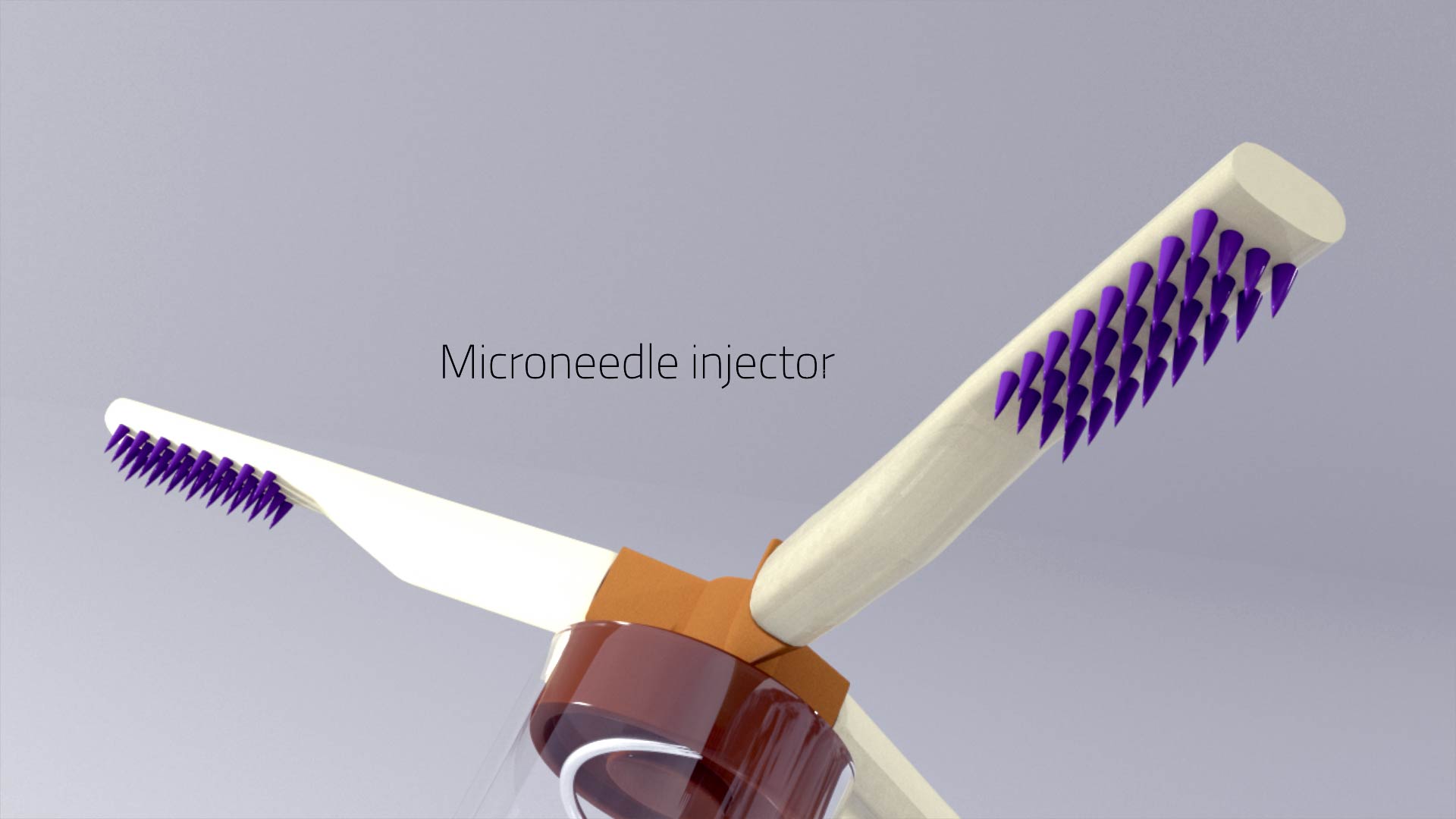
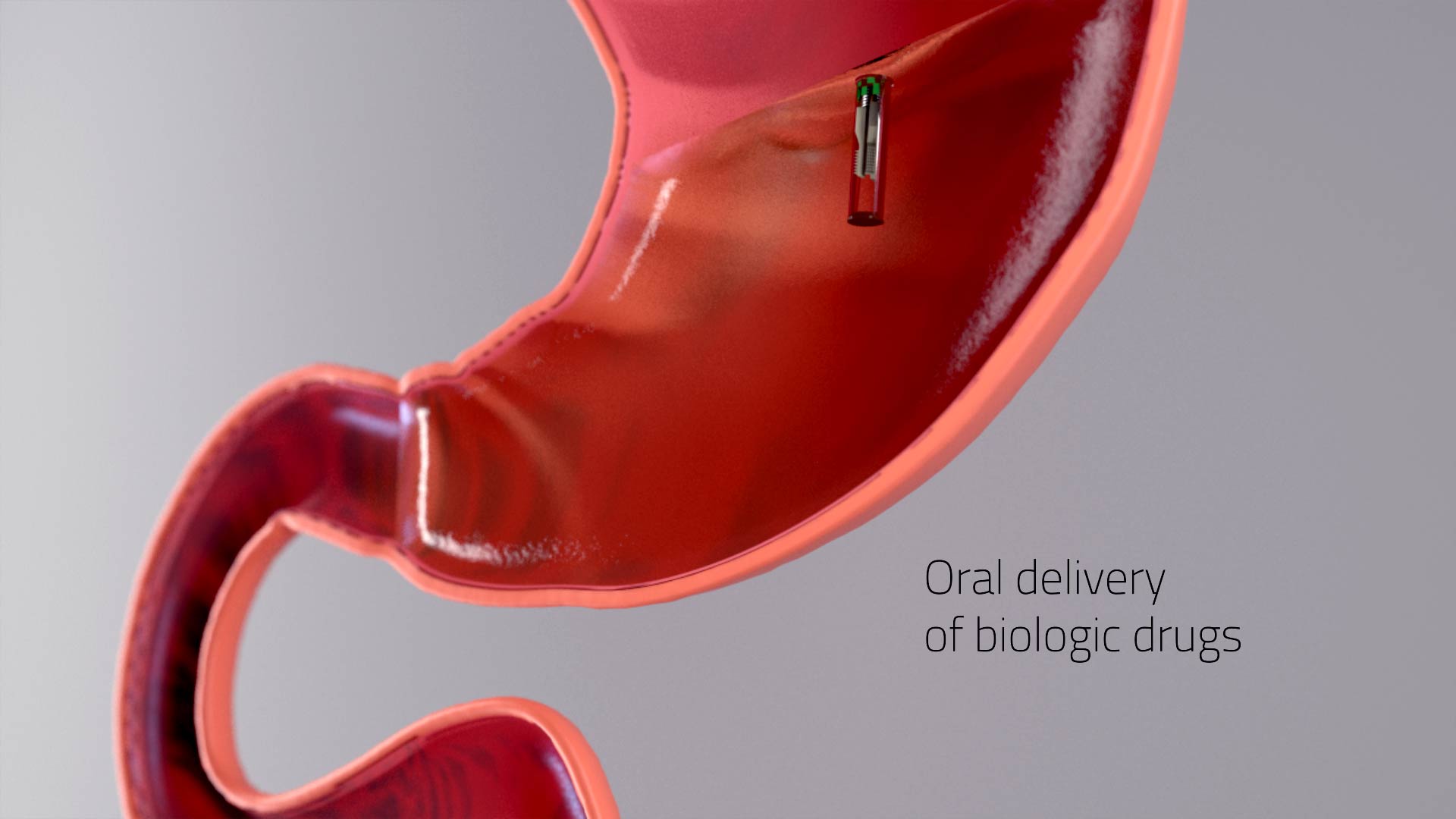

https://www.nature.com/articles/s41591-019-0598-9?fbclid=IwAR3SOhYka1bPkN6iyvlXdIO0qQV8CTkxNYONr584q2YThPKoni7aRPwz7s8
How to land a journal cover
 “Hers is a practical manual featuring simple techniques and easily accessible tools, such as flatbed scanners and smartphones, as well as advice on lighting, composition and resolution.”
“Hers is a practical manual featuring simple techniques and easily accessible tools, such as flatbed scanners and smartphones, as well as advice on lighting, composition and resolution.”
Source : How to land a journal cover
Cell Report cover project